An exploration into the concept of Firelocity!
By: Tony Wong, Jason Broome, and Mitch Thaler
Physics 2: period 3A
1/18/2007
Table of Contents
Introduction
Background Information: Why do things burn? What burns better than other things? What doesn’t burn? Our group is specifically concerned more with the question “What affects the speed of how fast fire travels?” It is obvious to see that fire moves faster on certain types of things. For instance, fire travels faster on gasoline than it does on a carpet. For our experiment, however, we want to see what makes fire travel faster. To do this experiment, we wanted to find a substance that was liquid and highly flammable. After searching for ideas, we chose isopropyl alcohol. Isopropyl is a highly flammable liquid, receiving a severe fire hazard warning of 3. Its atomic makeup consists of Carbon (3), Hydrogen (8) and Oxygen (1). It is colorless and smells similar to alcohol. A key idea is that isopropyl alcohol is soluble in water (OSHA). This is important because that is how we will alter the concentration of the alcohol. It is impossible to get 100% proof alcohol; the highest concentration that it can reach is 99%.
Question: Do different concentrations of isopropyl alcohol affect the rate at which it combusts? The concentration is a mixture of isopropyl alcohol and water. The rate we are measuring is the velocity. The independent variable is the different concentration levels. The dependent variable is the speed of combustion. The control variables are: the amount of solution that we combust, the length of the trough, the temperature of the trough, solution, and environment, and the time for the solution to vaporize.
Hypothesis: If there is a greater concentration of isopropyl alcohol (meaning the higher the percentage of isopropyl alcohol in an isopropyl/water solution), the fire will combust at a faster velocity because alcohol is highly flammable, and by diluting it, it will reduce its flammability.
Review of Literature: There are four major sources of ignition that we could use including: chemical, electrical, mechanical, and an open flame (Combustion Basics). We attempted to use an electrical spark to ignite the liquid vapors; however, we discovered it did not produce enough heat. Therefore, we decided a barbecue lighter that has an open flame would suffice. We also know that isopropyl alcohol will ignite at a flash point of 11.7 degrees (Johnson). The flash point is the lowest temperature that the vapors from the solution will ignite (Combustion Basics). Another important characteristic of isopropyl alcohol is the stoichiometric ratio. This is the ideal ratio of fuel to air in order to ignite a solution. If there is not a perfect ratio, then there is bad burn efficiency and flame instability (Stoichiometric Combustion). We have used the formula to balance the equation for isopropyl alcohol in order to solve for the ratio. The ratio is 1 mol of isopropyl alcohol to 4.5 mol of oxygen. The degree of heat that comes from the combustion is determined by how close we are to the perfect ratio. It is known that if 100% of the gas combusts, then it will produce the maximum amount of heat (Kumar). This leads us to wonder about the role of temperature with flammability.
(Back to Table of Contents)
Experiment
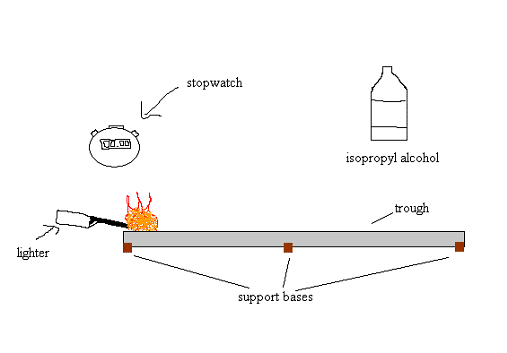
Procedure: First, assemble the trough with the base supports and check for levelness. Next, make the desired concentration. In order to do this, you must calculate the percentage of alcohol to water ratio out of 150 ml. Then measure the amounts with a graduated cylinder and mix them into a beaker. Here is a easy converting table:
|
Concentration
Percent |
Isopropyl
Alcohol amount (in ml) |
Water
amount (in ml) |
|
99% |
150 |
0 |
|
95% |
142.5 |
7.5 |
|
90% |
135 |
15 |
|
85% |
127.5 |
22.5 |
|
80% |
120 |
30 |
Once you have the desired concentration, pour the solution into the trough and let it vaporize for 1 full minute. After that, you are ready to ignite the solution. Start at one end of the trough and measure the time (in seconds) it takes for the flame to reach the other end. Record the data and repeat a total of 3 times for each concentration level.
Materials: Isopropyl alcohol, water, graduated cylinder and beaker, trough, base supports, and stopwatch.
Lab Setup: We purchased a metal trough (roofing material) which we measured to be 10 ft or 3.048 meters. We also decided on using 150 ml of solution in order to conserve the alcohol and create a good depth along the trough. The temperature of the room we tested in was about 60 degrees F. We were unable to get the humidity level. Our ignition starter is a typical barbecue lighter. We did not measure the amount of heat that this lighter produces. Should we repeat this experiment, it is important to document this temperature.

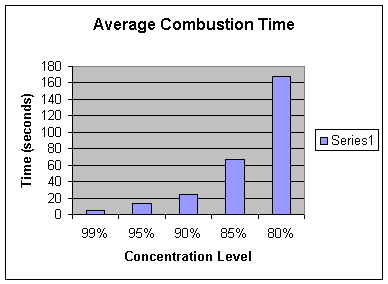
Combustion
Time
(Concentration
Level)
|
|
99% |
95% |
90% |
85% |
80% |
|
|
5.01 |
13.73 |
27.13 |
67.59 |
172.89 |
|
|
2 |
4.83 |
11.47 |
20.58 |
66.37 |
162.35 |
|
|
3 |
5.15 |
14.10 |
25.68 |
68.30 |
169.62 |
|
|
Average |
5.00 |
13.10 |
24.46 |
67.42 |
168.29 |
(Time
in seconds)
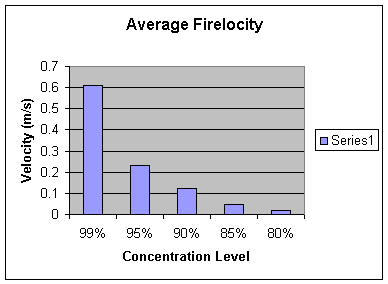
Calculated
Velocity of Combustion Rate
(Concentration
Level)
|
|
99% |
95% |
90% |
85% |
80% |
||
|
|
.608 |
.222 |
.112 |
.0451 |
.0176 |
||
|
2 |
.631 |
.266 |
.148 |
.0459 |
.0188 |
||
|
3 |
.592 |
.216 |
.199 |
.0446 |
.0180 |
||
|
Average |
.610 |
.233 |
.125 |
.0452 |
.0181 |
Scientific Thoughts
Analysis: Our hypothesis is proven correct. At a lower concentration the rate of combustion is significantly slower. For instance, the average velocity at 99% was .610 m/s compared to the average velocity at 80% which was .0181 m/s. Furthermore, the graph of the velocities shows an exponential decay as the concentration level decreases. For example, with each rise in the concentration level, the combustion time almost always doubled. Therefore, we believe the relationship between the concentration of the solution and the velocity of the fire is exponential. With that, we calculated the theoretical velocities of the fire for even lower levels of concentrations. The equation of an exponential line is y=a*b^x. The equation for this specific line is y= 1.782E -8* 1.189^x. Along with our hypothesis, mathematically we expect the fire to combust at 5.915E -4 m/s for 60%, 3.247 E-6 m/s at 30%, and 1.782E -8 m/s at 0%. Obviously, the solution will not burn at 0%. In fact, the solution may not even burn at 60% or 30%. With further experimentation, we could test at exactly what percentage of concentration does isopropyl alcohol not burn.
Another thing we noticed is that the temperature of the environment has a drastic effect on the combustion rate. We observed that outside where the temperature was much colder (about 40 degrees F) the solution did not ignite as well as inside a room at about 60 degrees F. If we were to conduct further exploration into firelocity, we would like to find the correlation between the rate of combustion and temperature. We hypothesize that the lower the temperature is, the slower the fire will travel across the isopropyl alcohol.
An interesting observation from the experiment is the physical behavior from the fire. As the flame spread down the trough, the solution was pushed along with it. We noticed a very small wave that formed at the front of the flame spread. Once the fire reached the end of the trough, we also saw that there was a build up of solution. We think this happened because the heat of the trough made the metal expand, pouring much of the solution to the bottom end. This, in turn, would affect the rate of combustion. As the heated solution mixes with the cold solution, it is much easier to ignite, causing a faster combustion rate.
Limitations: As always, human error contributed to inaccuracy. Because we used a stopwatch to measure the time of the flame spread, the time was not completely accurate. Another error is the levelness of the trough. It is extremely difficult to find a completely flat surface. Also after each trial, the temperature in the room began to rise. Consequently, the temperature of the vapors from the solution also rose, allowing for easier ignition. The only solution to keeping a closed environment consistent is to have the ability to manipulate temperature and humidity perhaps through the use of an air conditioner/fan. Also, because of the heat, the metal trough became warped with every trial. This in turn might have affected the speed of the flame spread. An easy solution would be to use a thicker, sturdier metal trough. Along with the trough, there was a small leak at both ends. This obviously decreases the amount of solution in the trough, affecting the amount of vapors in the air and the overall outcome. Another limitation is the fact that isopropyl alcohol does not exist at a 100% concentration level. This is due to the fact that the humidity of the air will always add moisture to the alcohol. The only way to get 100% is unknown to us. When we mixed the different concentrations, we always lost a small amount of the solution to the sides of the cylinder and sides of the trough. Along the sides of the trough, there was also a build up of carbon from the fire. There were also some particles that fell into the solution before it was ignited. These could have slowed down the speed of the fire. To fix this, we could either clean the trough each time or have a new trough for each experiment.
(Back to Table of Contents)
Works Cited
“Combustion Basics”. Seneca College. 2006. <http:/professorpyro.com/page1/page1.html>
: This was a good introduction to the concept and reactions
of different chemical combustions.
“Stoichiometric Combustion.” The Engineering Toolbox. 2005.
< http:/www.engineeringtoolbox.com/stoichiometric-combustion-d_399.html >
This helped us figure out the stoichiometric ratio for a
theoretically perfect combustion.
Johnson, Patrick L., and Randall, Eric. “Isopropyl Alcohol.” Paddock Laboratories, Inc 18 October 1991. 16 January 2003.
<http:/www.paddocklabs.com/images/ISO99PDF-MSDS.pdf>
This site gave us an in-depth description of the
properties of isopropyl alcohol.
Kumar, Sunil. “Combustion Basics.” September 2002.
<http:/www.energysolutionscenter.org/boilerburner/Workshop/Combustion.htm>
This site also described the basics of combustion while
giving a real life example of a flammable combustion burner.
“OSHA Guideline for Isopropyl Alcohol.” Occupational Safety and Health Administration. September 1996.
<http:/www.osha.gov/SLTC/healthguidelines/isopropylalcohol/recognition.html>
This site also gave us an in-depth description of the
properties of isopropyl alcohol and included information for safety.
(Back to Table of Contents)