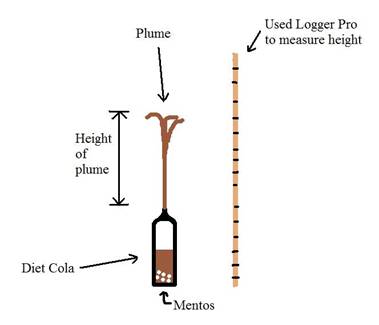
 |
Results
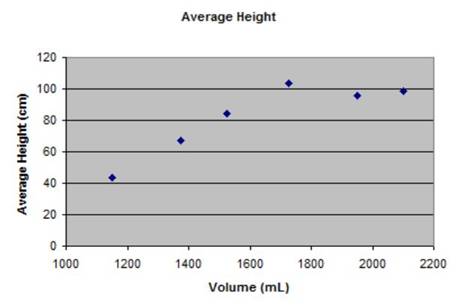 |
This graph shows all trials of heights, so you can see the outliers:
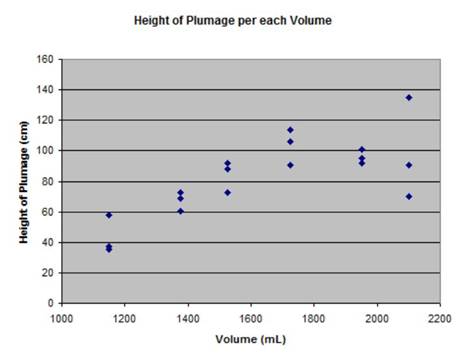 |
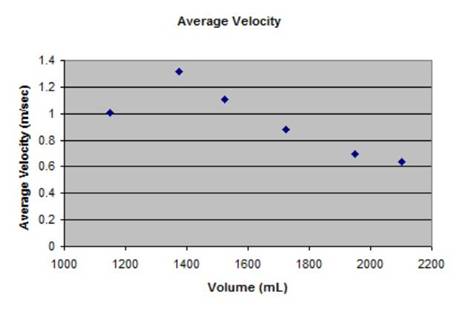 |
Data file: text .:. Excel
Calculation of velocity: Velocity = Distance/Time
As the data table and graphs show, the average peak height of plume was not when the bottle was full as we had predicted, but at 1725mL.
Uncertainties were found as follows: For volume, we used a measuring device that had intervals of 50mL, giving us an uncertainty of +/- 25mL. For height, our uncertainty was human error in Logger Pro, so it was not exact. We estimated +/- 3cm. For time, our video in Logger Pro had a time of 0.2 seconds between each slide, making our uncertainty +/- 0.1.
Conclusion
We have concluded that there is an optimal amount of soda to maximize the height of the foam explosion. It peaked at the second highest amount of soda, and we were surprised with the outcome. Our hypothesis was not supported, because there is an amount of soda between full and empty that creates a bigger plume. When we discovered the full soda did not achieve the same height as the trials with slightly less we were shocked. We had assumed that the more liquid there was the more aspartame and carbonation there would be to react. We could not explain this phenomenon but we theorize that the extra space in the bottle allowed more bubbles to form prior to the explosion, therefore providing a higher plume because the bubbles are more easily propelled than the liquid. However, there are many sources of error that could have altered our data. When the sodas were opened some of the reacting gases may have left the bottle at varying speeds, and there were inconsistencies in pouring out the soda as we attempted to do it quickly. Also, the mechanism we used to drop the Mentos was not perfectly efficient, resulting in Mentos that were not all dropped at the same time or speed. And finally some human errors such as shaky video and not exact measurements in Logger Pro. We could have improved the experiment by performing more trials for each amount of soda and by being more careful when following the steps of the method.
-
http://www.newscientist.com/article/dn14114-science-of-mentosdiet-coke-explosions-explained.html - This website provides a thorough explanation of the science behind the resulting explosion when diet cola and Mentos come in contact.
-
http://www.youtube.com/watch?v=9vk4_2xboOE - This video shows the potential of the diet cola and Mentos in an extreme domino effect.
-
http://dsc.discovery.com/videos/mythbusters-diet-coke-and-mentos.html - The MythBusters reveal the science behind the famously explosive cola-candy combo.
-
http://www.eepybird.com/featured-video/the-extreme-diet-coke-mentos-experiments/ - These guys spend hours creating tons of different experiments with diet cola and Mentos, providing us with exciting videos.
-
http://thetartan.org/2007/2/19/scitech/how - Further explanation of why diet cola and Mentos react with and explosion.
Works Cited
http://www.eepybird.com/featured-video/the-extreme-diet-coke-mentos-experiments/
http://www.newscientist.com/article/dn14114-science-of-mentosdiet-coke-explosions-explained.html
http://www.youtube.com/watch?v=9vk4_2xboOE
http://dsc.discovery.com/videos/mythbusters-diet-coke-and-mentos.html