Temperature VS. Viscosity
Reed Hall; IB Physics II
The history of maple syrup goes back hundreds of years. The Native American legend of the discovery of the sticky substance began when “the chief of a tribe threw a tomahawk at a tree, sap ran out and his wife boiled venison in the liquid” (Pickert). It takes about forty gallons of pure tree sap to make one gallon of maple syrup, since the sap is 98% water. In the early 18th century, dairy farmers used syrup as a substitute to selling milk. Maple syrup became a luxurious commodity, because it had such a small window to be farmed, “during the brief weather window between winter and spring” (Pickert). The maple syrup dripped slowly from the tree, due to the cooler temperatures. Nowadays, syrup is a staple to breakfast; what else beats the sugary goodness?
It is most likely true that one has noticed that syrup pours much easier and faster when it is heated, leading to the question about how temperature plays into the characteristics of a specific liquid. The viscosity of a liquid is defined as “the quantity that describes a fluid's resistance to flow. Fluids resist the relative motion of immersed objects through them as well as to the motion of layers with different velocities within them” (Elert). Possible variables that can impact the viscosity of a liquid are temperature, pressure, and flow conditions. Since there are a few factors impacting the viscosity of the liquid, I decided to focus solely on the effect that temperature has on the liquid. Viscosity changes when the temperature changes because the molecules within that liquid move much faster, allowing the flow to increase. The viscosity of a liquid is said to decrease when the temperature is raised, which ultimately changes the shear stress of the liquid. Similarly to maple syrup, the viscosity of different liquids can change when outside forces act upon it. This can happen because it changes the stress in the liquid, meaning it will flow differently when outside forces are acted upon it, such as temperature.
The purpose of this investigation is to determine the effect that temperature has on the viscosity of different liquids.
I believe that as temperature rises (with temperature of the liquid as the independent variables and the time it takes for a marble to travel a specific distance as the dependent variable) in a liquid, we can expect the marble to travel much faster than cooler liquid. This is because an increase in temperature increases the flow of a liquid, which makes it lose its’ viscosity. I aim to be able to calculate the viscosity of different liquids at different temperatures and to prove whether heat does play a role in the viscosity of that liquid. I have this hypothesis because
For my experiment, I was going to see how the difference in
temperature would affect the viscosity of the liquid. I was going to drop a marble
into a clear PVC pipe and time the distance it takes to travel from one end of
the pipe to another. I wanted to ensure validity so I did three drops and
averaged the times and velocities of the marbles. I would heat up a certain
liquid to a specific temperature, pour it into the pipe, do my drops, remove
the liquid from the pipe, heat up the liquid to my next desired temperature,
and repeat the same process. The process will be repeated for five different
liquids at four different temperatures ranging from 20-80 degrees celsius. I did not include 100 degrees celsius
because it would result in some of my liquids vaporizing or becoming
caramelized, meaning it would just skew my data.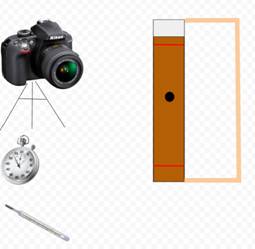
Since calculating the density of a specific liquid would be difficult, considering all the precise measurements required, I will be measuring the velocity of the marble as the temperature increases. While it does not directly measure viscosity, a change in velocity on the marble means a change in the density and the flow of a liquid (if the marble descends faster, the viscosity is lower). Velocity has a direct correlation between temperature and viscosity changes, so it would eliminate a source of error and miscalculations.
For my project, I tested five different liquids: water, vegetable oil, alcohol, shampoo, and maple syrup. I used a clear PVC pipe for clear, unrestricted viewing of marbles that I dropped into the liquids. I needed a camera in slow motion to view the descent of the marbles, a stopwatch, an accurate thermometer, colored bands, and a base for the PVC pipe to keep it level.
A diagram of my setup can be seen at right.
Independent Variable(s): Liquid that is heated up to a desired temperature, usually ranging from 20-80 degrees.
Dependent Variable(s): Time for marble to reach the bottom of the tube.
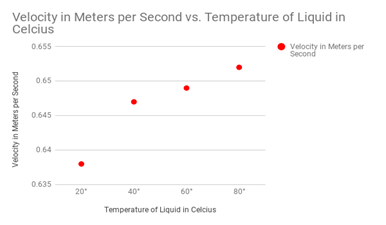
Firstly, the difference between the drops with water resulted in a different of velocity of .1359 meters per second between my 20° and 80° drops. Next, for my vegetable oil, the difference in velocity was .144 meters per second between 20° and 80°. Third, for shampoo, the difference between 20° and 80° was .5151 meters per second. This was my strongest case in argument for temperature affecting viscosity simply because the change was so massive. For maple syrup, the difference between 20° and 80° was .332 meters per second. Lastly, alcohol had a slight change of .014 meters per second.
The most important liquids that I wanted to test were the liquids with the thicker viscosity. These important liquids included the shampoo and the maple syrup. The liquids with the higher viscosity are much harder to prove because the flow does not differentiate much. These liquids included the water and the alcohol. A diagram for velocity of the shampoo drops can be seen at the right, notice how velocity changes substantially when it is heated up above 60 degrees celsius.
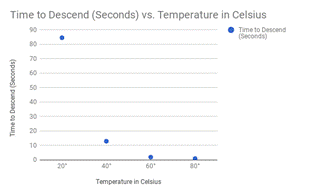
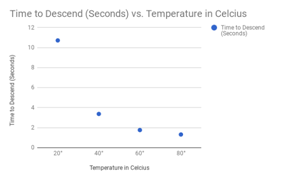
Here are my graphs for all of my tests:
Water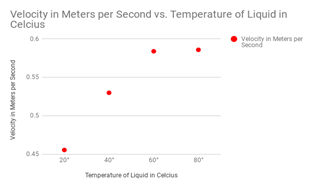
Vegetable Oil
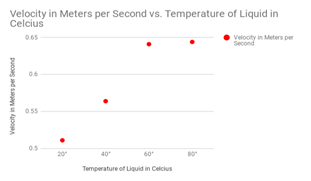
Shampoo
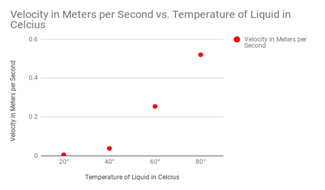
Maple Syrup
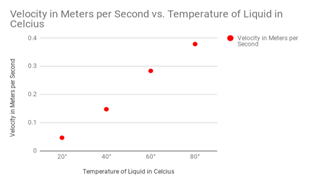
Alcohol
Link
to Text
Spreadsheets
Link to Excel Spreadsheet
Sources of Error
Perhaps, one of my largest sources of error included the measurement of temperature accurately. There are many things that went into the measuring and possible error for my measurements. It is difficult to keep a temperature accurate since heat is convected so quickly, so accurate temperature is major in avoiding a source of error. In addition, an inaccurate thermometer could have measured the temperature off by a fraction of a degree (I had an electric thermometer). There could also be an error in the timing of my drops, since I had to review footage in slow motion, I tried to remove a source of error by syncing my timer with the video, however, there is always a small margin of error. The last possible source of error could have been in a slight variation in weight of my three marbles that I dropped, but they all stayed within the area of 5.3 grams and the difference would have not had a major effect on my tests.
In conclusion, temperature does affect the change in velocity. This can be seen in the changes in velocity in trials that tested liquids with higher viscosities. The most convincing test was my shampoo test because it started off barely moving .006 meters per second to moving .521 meters per second. If I were to do this experiment again, I would focus on keeping the temperature controlled and trying to keep it from losing heat. I would also test liquids with higher viscosity. This happened because when I changed the temperature, the stress became less, which allowed the ball to travel much faster through the liquid. When a liquid is cooler, the stress is higher, and my marble did not travel as fast, this can be seen through my tests above. The surface tension, impacted by the changing of the temperature also changes, which allows the marble to travel through easier when temperature is raised.
Pickert, Kate. “A Brief History of Maple Syrup.” Time, Time, 16 Apr. 2009, time.com/3958051/history-of-maple-syrup/.
Elert, Glenn. “Viscosity.” Viscosity – The Physics Hypertextbook, The Physics Hypertextbook, 1998, physics.info/viscosity/.
Mexger, Thomas G. “Factors Affecting Viscosity.” Factors Affecting Viscosity, 12 May 2014, www.viscopedia.com/basics/factors-affecting-viscometry/.
Related Sites:
https://en.wikipedia.org/wiki/Viscosity
This is a website that explains how viscosity works, what it is, and how it affects the way liquids flow.
https://en.wikipedia.org/wiki/Maple_syrup
This is a website that explains the history of maple syrup. The introduction of this essay explained how viscosity of maple syrup affects the rate in which the syrup is farmed.
https://www.azom.com/article.aspx?ArticleID=10036
This is a website that explains how temperature impacts viscosity. This is effective at giving a totally “dumbed down” explanation of how this works.
https://www.engineeringtoolbox.com/boiling-points-fluids-gases-d_155.html
This is a website that explains how different liquids have different boiling points, which can impact the viscosity of that said liquid. This is important because I had to choose what my final temperature would be for each liquid.
https://sciencing.com/changing-temperature-affect-viscosity-surface-tension-liquid-16797.html
This website further explains how the changing of temperature impacts the viscosity of a liquid. It backs up my research question, since a community of scientists worked on this and not one high school student.